Hydrogen sulphide (H₂S) is one of the most critical contaminants present in raw biogas produced from anaerobic digestion. Although it is typically present in trace concentrations compared to methane and carbon dioxide, its impact on plant reliability, safety, and lifecycle cost is disproportionately high. H₂S is a toxic, corrosive, and chemically reactive gas, making it one of the most problematic impurities for any biogas-to-CBG conversion system.
H₂S is a weak acid gas with high affinity toward metals, especially iron and copper-based alloys. Its behavior becomes more aggressive in the presence of moisture, elevated temperature, and oxygen traces—conditions commonly found in biogas pipelines, compressors, and upgrading units. Unlike CO₂, which mainly affects gas calorific value, H₂S actively destroys plant hardware and threatens human safety.
Another challenge with H₂S is that it is not linear in impact. Even small increases in concentration can drastically accelerate corrosion rates, poison catalysts, and damage adsorption media. This makes precise monitoring and control absolutely essential in CBG plants.
Biochemical Origin of H₂S in Anaerobic Digestion
Role of Sulphur in Feedstock
All organic feedstocks used in CBG plants—such as agricultural residues, press mud, poultry litter, food waste, and sewage sludge—contain sulphur in various forms. Sulphur is naturally present in:
- Proteins (as sulphur-containing amino acids like cysteine and methionine)
- Sulphates
- Organic sulphur compounds
When these materials enter the anaerobic digester, sulphur becomes part of the microbial conversion process.
Sulphate-Reducing Bacteria (SRB) Activity
Within the digester, a specific group of microorganisms called Sulphate-Reducing Bacteria (SRB) competes directly with methane-forming bacteria. SRBs use sulphates as electron acceptors and convert them into sulphides during metabolism. These sulphides then react with hydrogen ions to form hydrogen sulphide gas.
This process is problematic because:
- SRBs consume substrates that methanogens need
- Increased SRB activity reduces methane yield
- More sulphur ends up in the gas phase as H₂S
High sulphur feedstock or poor digester control (pH imbalance, temperature fluctuations) significantly accelerates this mechanism.
Influence of Digester Operating Conditions on H₂S Formation
Hydrogen sulphide generation in a CBG plant is strongly governed by digester operating conditions, not just by feedstock sulphur content. Even with the same raw material, poor control of digestion parameters can increase H₂S concentration by several times.
1. Effect of pH on H₂S Release
pH plays a decisive role in determining whether sulphur remains dissolved in the slurry or escapes into the biogas phase.
Inside the digester, sulphur exists in equilibrium between:
- Dissolved sulphide ions (HS⁻, S²⁻)
- Gaseous hydrogen sulphide (H₂S)
At neutral to slightly alkaline pH (7.2–7.8), most sulphur remains in ionic form within the digestate. However, when pH drops:
- The chemical equilibrium shifts
- Sulphide ions recombine with hydrogen ions
- Free H₂S gas is released rapidly into biogas
This is why acidic digestion conditions immediately cause H₂S spikes, even if sulphur input remains constant.
2. Effect of Temperature on H₂S Formation
Temperature affects H₂S generation through microbial activity and reaction kinetics.
At higher temperatures:
- Sulphate-reducing bacteria (SRB) become more metabolically active
- Biochemical reactions occur faster
- Sulphate reduction rate increases
In thermophilic digesters or poorly controlled mesophilic systems:
- SRB growth can outpace methanogens
- More sulphur is converted to sulphide
- H₂S production accelerates
H₂S Removal Techniques
In-Digester H₂S Reduction (Primary Control Method)
In-digester methods aim to prevent H₂S formation or release at its source, thereby reducing stress on downstream gas-cleaning equipment. These techniques do not produce pipeline-grade gas by themselves, but they significantly improve overall plant stability and economics.
Micro-Aeration
Working Principle
Micro-aeration involves injecting a very small, carefully controlled quantity of air or pure oxygen into the digester headspace or recirculation line. This oxygen is not meant for combustion; instead, it supports the growth of sulphur-oxidizing bacteria (SOB).
These bacteria convert dissolved sulphides (HS⁻) into elemental sulphur (S⁰) according to the reaction:
H₂S + ½ O₂ → S⁰ + H₂O
The elemental sulphur remains in the digestate or deposits on digester surfaces, preventing it from entering the biogas phase.
Advantages
- Reduces H₂S at source (30–60%)
This reduction stabilizes H₂S fluctuations and protects downstream systems. - Low capital cost
Only requires controlled oxygen dosing, blowers, and instrumentation. - Reduces load on downstream systems
Smaller scrubbers, longer media life, lower OPEX
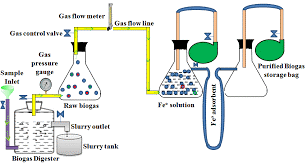
Dry Scrubber
In dry scrubbers, biogas passes through a fixed bed of iron-based media. H₂S reacts chemically with iron oxide:
Fe₂O₃ + 3H₂S → Fe₂S₃ + 3H₂O
The sulphur is chemically bound to the media until it becomes saturated and must be replaced or regenerated
Activated Carbon-Based H₂S Removal
Working Principle
Activated carbon impregnated with chemicals (KOH, KI, metal salts) removes H₂S via adsorption + chemical reaction. H₂S molecules are trapped inside the porous carbon structure and converted into stable sulphur compounds.
Advantages
- Achieves very low outlet H₂S (<1 ppm)
Essential for PSA, membrane, and CBG bottling. - Compact system
Small footprint with high efficiency.
Biological Scrubber / Biofilter
Working Principle
Biological scrubbers use sulphur-oxidizing bacteria immobilized on packing media. Under controlled oxygen conditions, bacteria convert H₂S into elemental sulphur or sulphate.
This process combines mass transfer + biological oxidation, making it suitable for continuous large-scale operation.
Advantages
- Low chemical usage
Bacteria perform the conversion. - Environmentally friendly
Minimal hazardous waste. - Suitable for high and continuous H₂S loads
Chemical Scrubbing (Caustic / Oxidative)
Working Principle
H₂S is absorbed into alkaline or oxidizing solutions such as NaOH, NaOCl, or H₂O₂, forming stable sulphur salts.
Advantages – Explained
- Very high removal efficiency
- Handles extremely high H₂S concentrations
- Rapid response to load variations
Conclusion
Hydrogen sulphide removal is a critical challenge in the design and operation of CBG plants. Its corrosive nature and toxicity make it imperative to address H₂S not as a mere contaminant, but as a system-wide risk affecting digester biology, gas quality, equipment durability, safety, and overall plant economics.
A successful H₂S management strategy requires a multi-tiered approach starting with in-digester biological and chemical control methods such as micro-aeration and iron salt dosing to reduce sulphide formation at the source. This reduces the burden on downstream removal systems, which often combine dry scrubbers, activated carbon polishing, biological scrubbers, or chemical scrubbing to meet stringent gas quality requirements.
Equally important is the selection of corrosion-resistant materials and robust operational controls to withstand the harsh chemical environment created by H₂S and moisture. Ignoring material compatibility or relying solely on downstream treatments leads to frequent equipment failures, safety hazards, and costly downtime.